 This structure is referred to as the zinc-blende lattice, named after. Rock candy, the crystalline form of table sugar or sucrose, is an example of a molecular crystal. Each atom still has four covalent bonds, but these are bonds to atoms of the other type. Molecular crystals tend to be soft with relatively low melting points. A molecular crystal is held together by non-covalent interactions, like van der Waals forces or hydrogen bonding. Molecular Crystals: These crystals contain recognizable molecules within their structures.Table salt (NaCl) is an example of this type of crystal. Ionic crystals are hard and have relatively high melting points. Ionic Crystals: The atoms of ionic crystals are held together by electrostatic forces (ionic bonds).Metal atoms can pack in hexagonal closest. Metallic crystals tend to be very dense and have high melting points. The structures of crystalline metals and simple ionic compounds can be described in terms of packing of spheres. This leaves the outer electrons of these atoms free to float around the lattice. Metallic Crystals: Individual metal atoms of metallic crystals sit on lattice sites.Examples of covalent crystals include diamond and zinc sulfide crystals. Many covalent crystals have extremely high melting points. You can think of a covalent crystal as one big molecule. Covalent Crystals: A covalent crystal has true covalent bonds between all of the atoms in the crystal.LatticeA = go.Scatter(visible = True,x=pattern_points.T,y=pattern_points. Pattern_points = lattice_generation(a1,a2) Return dash_trace(dotLine_a1,color),dash_trace(dotLine_a2,color)ĭef make_arrow(vec,text,color = 'Red',vec_zero =, text_shift = ): # Produces the dotted lines of the unit cellĭef dash_contour(a1,a2, vec_zero = np.array(), color='Red'):ĭotLine_a1 = np.transpose(np.array()+vec_zero)ĭotLine_a2 = np.transpose(np.array()+vec_zero) # Produces the lattice to be fed into plotly In the image below we show a 2D simple square lattice (panel A).īecause the environment of each point is the same, this set of points forms a lattice. With a i being the primitive lattice vectors.įor a n dimensional system, we have to define n linearly independent primitive lattice vectors in order to be able to map out the entire lattice.īut there exist multiple equivalent definitions of a lattice.Ī lattice is a set of points, where the environment of each point is the same. R = n 1 a 1 + n 2 a 2 + n 3 a 3, for n 1, n 2, n 3 ∈ Z. 
Such a framework is given by the concept of a lattice:Ī lattice is an infinite set of points defined by integer sums of a set of linearly independent primitive lattice vectors(will be explained later on). To describe such periodic structures, we need a framework. Most of solid state physics deals with crystals, which are periodic multi-atomic structures.  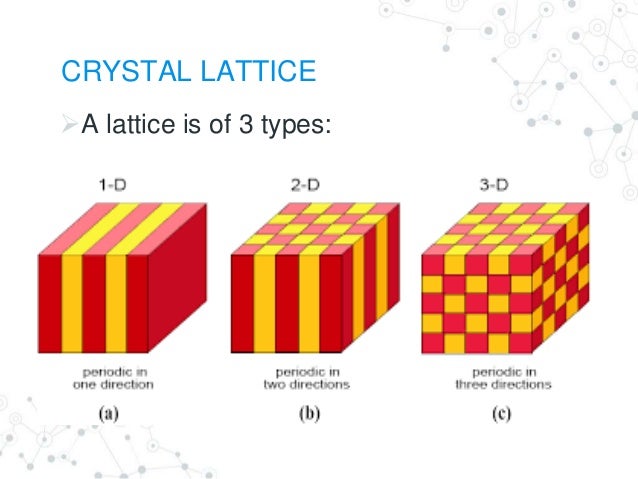
Therefore the upcoming two lectures will focus on developing, understanding, and applying this language and framework. It does however, require a new language and framework to fully describe such structures. The systems we considered were mainly 1D.īut most solids, such as crystals, are 3D structures.ĭescribing 3D systems is not much harder than describing 1D systems. In the past few lectures, we derived some very important physical quantities for phonons and electrons, such as the effective mass and the dispersion relation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
